One of the major issues pharmaceutical company Eli Lilly (LLY) has faced in recent days is meeting the demand for its Zepbound weight loss drug. However, an updated version of Zepbound may actually help on that front, and Eli Lilly shares are up fractionally in Tuesday afternoon’s trading.
Claim 30% Off TipRanks
- Unlock hedge fund-level data and powerful investing tools for smarter, sharper decisions
- Discover top-performing stock ideas and upgrade to a portfolio of market leaders with Smart Investor Picks
Zepbound will now be available in 2.5-milligram and 5-milligram single-dose versions for $399 per month and $549 per month, respectively. Normally, users take a 2.5-milligram version at first and eventually switch to higher doses before ultimately transitioning to a lower maintenance dose. Lilly is also selling these doses on a direct-to-consumer basis in an attempt to reduce markups, which typically push the price up to $1,000 a month.
Still, the biggest difference is that, normally, Zepbound comes in an autoinjector pen. However, the new version comes in vials that need to be manually inserted and injected from a standard syringe. This will help relieve some supply problems, as the autoinjector pens are proving to be the bottleneck here, and regular vials are much easier to produce.
Fighting the Knockoffs
The news comes as a disaster to several companies who were working to fill the gap with a variety of solutions, ranging from compounded medications—which are modestly risky depending on where they are found—to other versions, which have much less quality control and are, therefore, more dangerous overall. But these knockoffs and copycats had one critical advantage: they were available.
It really does not matter how good a drug is if it cannot be bought at any price. This move from Lilly helps ensure it will capture the maximum market possible and, ultimately, win from there.
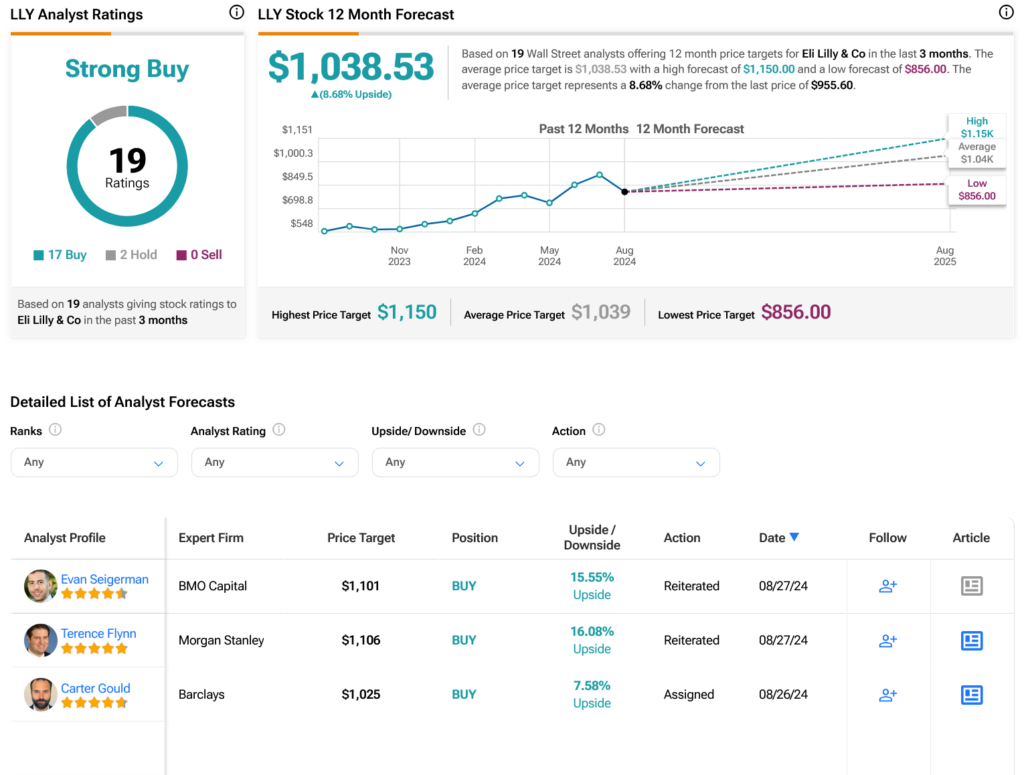
Is Eli Lilly a Buy, Sell, or Hold?
Turning to Wall Street, analysts have a Strong Buy consensus rating on LLY stock based on 17 Buys and two Holds assigned in the past three months, as indicated by the graphic below. After a 73.2% rally in its share price over the past year, the average LLY price target of $1,038.53 per share implies 8.68% upside potential.