Shares of biopharmaceutical company Synlogic (NASDAQ:SYBX) are on the rise today after its product candidate SYNB1934 bagged an orphan drug designation from the U.S. Food and Drug Administration for the treatment of phenylketonuria (PKU).
Claim 30% Off TipRanks
- Unlock hedge fund-level data and powerful investing tools for smarter, sharper decisions
- Discover top-performing stock ideas and upgrade to a portfolio of market leaders with Smart Investor Picks
Next, Synlogic is planning to begin a Phase 3 trial of the drug in the first half of 2023. Impressively, SYNB1934 has also bagged a Rare Pediatric Disease designation in the U.S. as well as an orphan drug designation in Europe.
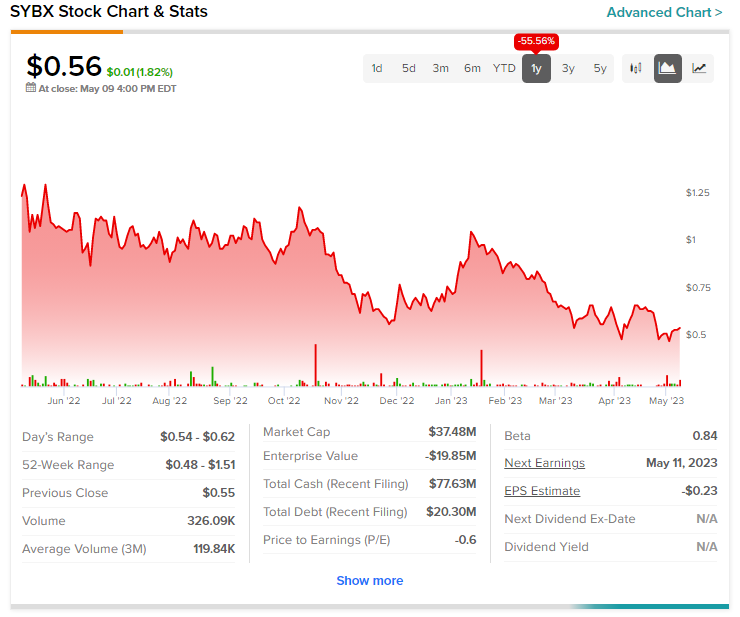
Today’s price gains come after a nearly 55.5% drop in Synlogic shares over the past year.
Read full Disclosure


